Detailed
China is a big country with liver disease. According to conservative estimates, the number of liver patients is as high as 400 million, about 93 million are infected with hepatitis B virus, 13 million are infected with hepatitis C virus, 60 million are alcoholic fatty liver, and 200 million are non-alcoholic fatty liver. The huge patient base has seriously reduced the quality of my country's population and increased the national economic burden, which has restricted our country's progress toward the ranks of developed countries. Thoroughly improving the current status of liver disease in my country has become an urgent problem that people's public servants want to solve. By increasing the support of basic research, new drug research and development of liver disease. It has made China one of the most active countries in the world for liver disease research and development. As a cell model, primary hepatocytes play an irreplaceable role in the basic research of liver disease and the development of new drugs. At the same time, primary hepatocytes, as a living carrier of liver function, have important application value in the medical fields such as hepatocyte transplantation, bioartificial liver and 3D printing of liver organs.
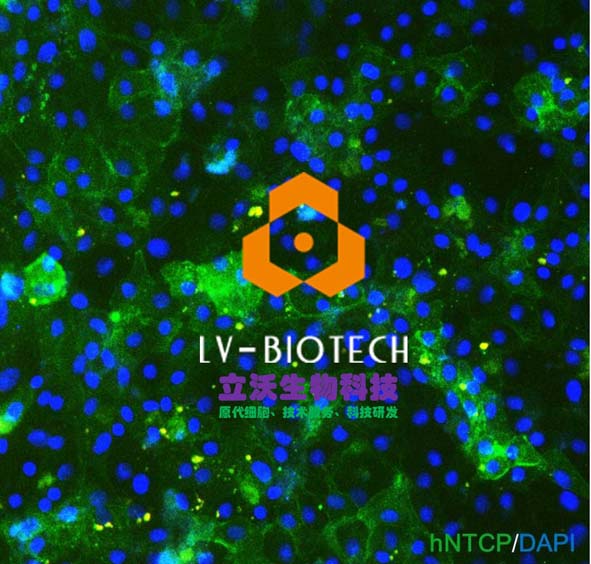
Primary hepatocytes refer to hepatocytes cultured immediately after being taken out of the body. Compared with traditional liver cancer or immortalized cell lines, they have highly differentiated characteristics and are closest to the real cell environment in the body. They are liver metabolism, liver disease, and hepatitis virus. "Gold Standard" cell model in other fields. The FDA and CFDA stipulate that in the "Technical Guiding Principles of Drug Nonclinical Pharmacokinetics Research", the importance of primary human hepatocytes and their accessory products in drug development is clearly defined. Primary hepatocytes have a complete drug absorption and metabolism system, including enzymes related to drug transport (such as NTCP), toxicity (such as MPR2, etc.), and metabolism (such as CYP450 series enzymes), which can best reflect the true response of cells to drugs. In particular, the expression levels of genes related to drug metabolism vary greatly among different races, and the development of drugs targeting Chinese people requires primary liver cells with Chinese genetic background to obtain accurate drug safety data. Therefore, primary hepatocytes and their ancillary products are just needed in the process of research and development of new drugs, helping the rapid development of biomedicine in my country.
Primary human hepatocytes are the smallest living unit of liver function and have important clinical application value. Primary hepatocytes mainly include three clinical application scenarios. Scenario 1: Orthotopic liver transplantation is waiting for the "life bridge" of donor liver. Every year, about 400,000 patients in China need liver transplantation. There are only about 4,000 patients undergoing liver transplantation, and about 90% of patients are waiting for donor liver. Died during the process; hepatocyte transplantation can effectively extend the patient's waiting time until a successful liver transplantation. Scenario 2: There are more than 1 million new cases of acute and chronic liver failure in China every year. Most patients rely on traditional artificial liver, which has poor treatment effect and many deaths. The primary hepatocyte are used to prepare "biological artificial liver", which is related to liver cells in vivo The physiological function is the closest, and the therapeutic effect is theoretically better. It is expected to iterate the traditional artificial liver. Scenario 3: Hepatic cells will be transfused to treat liver metabolic genetic diseases; China has a large population base and a large variety of liver metabolic genetic diseases, including birth defects of the A urea cycle (fatal, with an incidence of 1:30,000 to 1:100,000), Crigler–Najjar syndrome type I (fatal, 1:100,000), glycogen storage disease type I (fatal, 1:100,000 to 1:300,000), factor VII deficiency [symptom] (abnormal blood clotting, rare ), familial hypercholesterolemia (cardiovascular disease, incidence rate as high as 1/500), hereditary tyrosinemia type I, caused by FAH gene defects (liver failure, cirrhosis and hepatocellular carcinoma (HCC) , Incidence 6.7: 100,000) and so on. In foreign countries, using primary hepatocytes for life-transplantation of liver transplantation, treatment of acute and chronic liver failure, and treatment of liver genetic metabolic diseases are mature treatment methods. Only domestic 302 hospitals of the People's Liberation Army have done and reported relevant clinical trials, most of the cases All show the safety and effectiveness of liver cell transplantation.
Human hepatocyte expansion technology based on FAH gene-deficient rat model is a necessary method to solve the problem of primary human hepatocyte origin. At present, primary human hepatocytes are mainly derived from human donated materials and surgical removal of liver tissue. Due to Asian cultural differences, medical ethics, laws and regulations, it is very difficult to obtain primary human hepatocytes. The technology to be developed in this project is expected to completely solve the problem of primary human hepatocyte origin and avoid serious medical ethics issues. The expansion or regeneration technology of human hepatocytes based on animal models is an important way to standardize the production of primary human hepatocytes. primary human hepatocytes have large batch differences, especially the CYP gene related to drug metabolism, the ethnic difference is ten times, the individual difference can be up to thousand times, so the primary human hepatocytes used for drug metabolism test Donors mixed together. In spite of this, when some batches of primary human hepatocytes are used up, a new batch of mixed cells is still required. The new batch of mixed hepatocytes is unavoidable compared to the previous batch of cells Big difference. The human hepatocyte amplification technology based on the FAH gene-deficient rat model can theoretically obtain a steady supply of human hepatocytes. Primary human hepatocytes from multiple donors can be mixed or transplanted separately to establish a standard and stable primary human liver cell product are expected to become the "study object" in the industry.
Relevant articles published